First published in PTQ Q2 2021
Clément Salais, Associate Group Manager, Gas Business Group, Axens
In a typical steam methane reforming (SMR) unit designed to produce high purity hydrogen, methane reacts with steam in a dedicated heater to produce a converted syngas which, after CO shift, contains mainly hydrogen and CO2. The syngas is then purified through a PSA to produce hydrogen at 99.9% purity and a purge gas that contains CO2, CO, and some hydrogen. This purge gas is routed back as a fuel to the SMR furnace. The SMR furnace produces a flue gas containing all of the CO2 emitted by the SMR unit.
There are two main locations at which CO2 can be captured: either the converted syngas identified as location A or the flue gas identified as location B in Figure 1. CO2 absorption from syngas is much more favourable as it is at high pressure and there is consequently a large driving force to absorb CO2. The syngas is quite pure and does not contain any oxygen but it contains only part of the CO2 emitted by the SMR unit (up to 60% depending on the scheme and operating conditions).
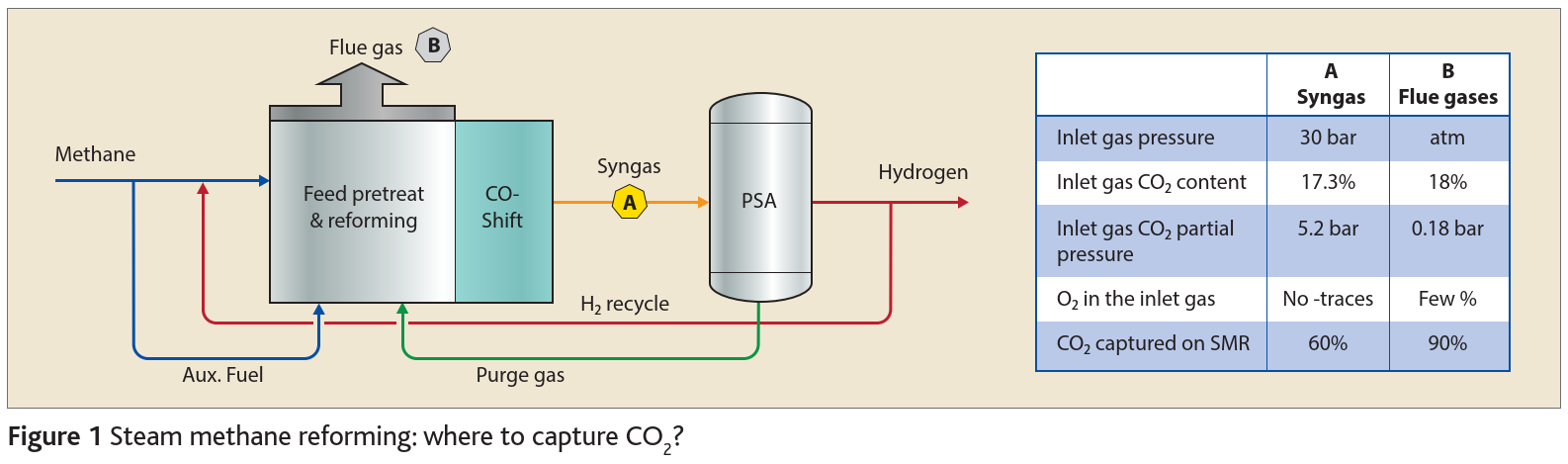
Adversely, the flue gas contains all of the CO2 emitted by the SMR and up to 90% of the CO2 emitted can be absorbed there. But the flue gas conditions are harsh: low pressure, high temperature, and a few per cent O2 in the gas.
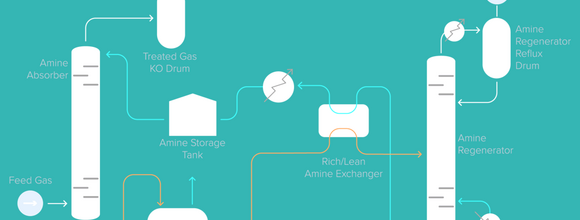
In terms of technology, a typical amine based acid gas removal unit with activated MDEA can be used to absorb all of the CO2 in syngas (up to 99%). Steam energy consumption can be reduced to about 1 GJ/t of CO2 with an optimised process scheme which avoids about 57% of the CO2 emissions of the SMR unit. The avoided CO2 readily takes into consideration emissions of CO2 due to energy consumption in the carbon capture unit. This gives a better insight into the amount of CO2 that is effectively recovered in the overall process.
CO2 emissions of a SMR unit can be significantly reduced by installing mature technology such as AdvAmine™ EnergizedMDEA technology which is licensed by Axens for syngas.
AdvAmine™
On the other hand, solvent based post-combustion carbon capture processes that are required to capture the CO2 in flue gas are much more energy demanding. The steam energy consumption of a first generation carbon capture process such as a MEA solvent based process is up to 3.7 GJ/t of CO2. The amount of CO2 avoided is, as a consequence, only 67% considering the additional CO2 emitted to regenerate the solvent. Post-combustion carbon capture from flue gas enables further reduction of CO2 emissions from the SMR unit but requires the development of new solvents and new processes to be more energy effective.
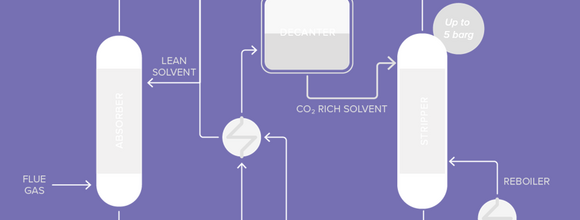
Axens is currently developing, with IFP Energies nouvelles, a new process for post-combustion carbon capture called the DMX process that is much less energy intensive than first generation processes. It is a solvent based technology but with a new solvent that has specific demixing capabilities under certain conditions of pressure and temperature. The energy consumption of the DMX process can be reduced to 2.3-2.9 GJ/t CO2 compared to the 3.7 GJ/t CO2 of the MEA process, leading to further reduction of CO2 emissions from the SMR unit as the CO2 avoided increases to 72% (see Table 1).

Its performance is proven at laboratory scale but needs to be demonstrated industrially in order to be ready for commercialisation. This demonstration is in progress through the 3D Project funded by the European Union (H2020 - Grant Agreement N°838031). It includes the construction of a demonstration unit at the ArcelorMittal Steel Mill in Dunkirk, France which is already in progress. Operation of the unit will begin in early 2022.
DMX™
Get the latest news about Carbon Capture, Utilization & Storage (CCUS)
Subscribe to our business newsletter to keep up to date with Axens news.
This newsletter gives access to our experts' insights and the opportunity to stay up-to-date with our latest products, technologies and services.